Difference between revisions of "Robertisla"
Robertisla (talk | contribs) |
Robertisla (talk | contribs) |
||
| Line 469: | Line 469: | ||
===Optimised Ion and rotatable molecule for 1-methyl-imidazolium chloride (HMim-Cl) A & B=== | ===Optimised Ion and rotatable molecule for 1-methyl-imidazolium chloride (HMim-Cl) A & B=== | ||
{| | {| | ||
| − | |[[File:IR_IMIDA_C_OPT_LABEL.png|300px]] ||<jmol><jmolApplet> | + | |[[File:IR_IMIDA_C_OPT_LABEL.png|300px]] ||===Jmol rotateable molecule=== |
| + | logfile: [[Media:IROBERTSON_NH3_OPTF_POP.LOG]] | ||
| + | <jmol><jmolApplet> | ||
| + | <title>optimised NH<sub>3</sub> molecule</title> | ||
<color>#7acccf</color> | <color>#7acccf</color> | ||
<size>250</size> | <size>250</size> | ||
| − | <uploadedFileContents> | + | <uploadedFileContents>IROBERTSON_NH3_OPTF_POP.LOG</uploadedFileContents> |
</jmolApplet></jmol> | </jmolApplet></jmol> | ||
| + | |||
|} | |} | ||
logfile: [[Media:IR_IMIDA_A&B_OPT_ION.LOG]] | logfile: [[Media:IR_IMIDA_A&B_OPT_ION.LOG]] | ||
Revision as of 10:31, 13 May 2026
Contents
- 1 Lab1 Marking
- 2 BH3 Molecule
- 3 BH3NH3 Molecule
- 4 Me3NH-CL Molecule
- 5 1-methyl-imidazolium chloride A
- 6 1-methyl-imidazolium chloride B
- 7 1-methyl-imidazolium chloride C
- 8 Ionic liquid ion pair (1-methyl-imidazolium chloride (HMim-Cl) A, B, and C)
- 9 NH3 Molecule
Lab1 Marking
You did a great job especially with the formatting. However, you have tabulated wrong charges for N and F. If you have any queries, please contact Prof. Hunt.
BH3 Molecule
Calculation data
| name of submitted log file | IR_BH3_OPTF_POP.LOG |
| molecule | NH3 |
| method | RB3LYP |
| basis set | 6-31G(d,p) |
| final energy | -26.615112 |
| RMS gradient | 0.003053 |
| point group | D3h |
Item Table
Item Value Threshold Converged? Maximum Force 0.000004 0.000015 YES RMS Force 0.000003 0.000010 YES Maximum Displacement 0.000017 0.000060 YES RMS Displacement 0.000011 0.000040 YES
Low frequencies data
| Low frequencies | -11.6940 | -11.6861 | -6.5543 | 0.0007 | 0.0280 | 0.4289 |
| Low frequencies | 1162.9745 | 1213.1390 | 1213.1392 |
Optimised molecule image
Jmol rotateable molecule
logfile: Media:IR BH3 OPTF POP.LOG
optimised BH molecule |
Important geometric parameters
Optimised bond distance and angle for BH3
r(B-H)=1.19Â
θ(H-B-H)=120°
Vibrational data
| Mode | 1 | 2 | 3 | 4 | 5 | 6 |
| Wavenumber (cm-1) | 1163 | 1213 | 1213 | 2583 | 2716 | 2716 |
| Symmetry | A2" | E' | E' | A1' | E' | E' |
| Intensity (arbitrary) | 93 | 14 | 14 | 0 | 126 | 126 |
BH3NH3 Molecule
Calculation data
| name of submitted log file | IR_NH3BH3_OPT.LOG |
| molecule | NH3BH3 |
| method | RB3LYP |
| basis set | 6-31G(d,p) |
| final energy | -83.22489 |
| RMS gradient | 0.000001 |
| point group | C1 |
Item Table
Item Value Threshold Converged? Maximum Force 0.000001 0.000015 YES RMS Force 0.000001 0.000010 YES Maximum Displacement 0.000043 0.000060 YES RMS Displacement 0.000019 0.000040 YES
Low frequencies data
| Low frequencies | -5.0461 | -2.8839 | -0.0019 | -0.0009 | -0.0005 | 0.6099 |
| Low frequencies | 263.3824 | 632.9842 | 638.4292 |
Optimised molecule image
Jmol rotateable molecule
logfile: Media:IR_NH3BH3_OPT.LOG
optimised NHBH molecule |
Important geometric parameters
Optimised bond distance and angle for NH3BH3
r(B-H)=1.210Â
r(N-H)=1.018Â
r(N-B)=1.668Â
θ(H-B-H)=114°
θ(H-N-H)=108°
θ(H-N-B)=111°
θ(H-B-N)=105°
Vibrational data
| Mode | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| Wavenumber (cm-1) | 263 | 633 | 638 | 638 | 1069 | 1069 | 1196 | 1204 | 1204 | 1329 | 1676 | 1676 | 2472 | 2532 | 2532 | 3464 | 3581 | 3581 |
| Symmetry | A | A | A | A | A | A | A | A | A | A | A | A | A | A | A | A | A | A |
| Intensity (arbitrary) | 0 | 14 | 4 | 4 | 41 | 41 | 109 | 3 | 3 | 114 | 28 | 28 | 67 | 231 | 231 | 3 | 28 | 28 |
Total energies
E (NH3) = -56.557769
E (BH3) = -26.615112
E (BH3NH3) = -83.22489
Association Energy
ΔE= E(NH3BH3)-[E(NH3)+E(BH3)]
ΔE= -0.051989 Au
ΔE= -136.49712 Kj/mol (5 d.p.)
Me3NH-CL Molecule
Calculation data
| name of submitted log file | IR_ME3NH-CL_OPT2.LOG |
| molecule | Me3NH-Cl |
| method | RB3LYP |
| basis set | 6-31G(d,p) |
| final energy | -26.615112 |
| RMS gradient | 0.003053 |
| point group | D3H |
Item Table
Item Value Threshold Converged? Maximum Force 0.000019 0.000450 YES RMS Force 0.000005 0.000300 YES Maximum Displacement 0.001834 0.001800 NO RMS Displacement 0.000343 0.001200 YES
Low frequencies data
| Low frequencies | -13.7046 | -2.3785 | -0.0021 | -0.0019 | 0.0012 | 1.7651 |
| Low frequencies | 55.6029 | 56.2004 | 189.7955 |
Optimised molecule image
Jmol rotateable molecule
logfile: Media:IR_ME3NH-CL_OPT2.LOG
optimised MeNH-Cl molecule |
Important geometric parameters
r(N-H)= 1.16395
r(H-Cl)= 1.73817
r(N-Cl)= 2.90211
Rigid Scan for Me3NH-Cl
PES Raw data plot
PES raw data table
| Scan Coordinate  | Total Energy (Hartrees) | Relative Total Energy kJ mol -1 |
| 0.8000000000 | -632.0662501520 | 229.8654262 |
| 0.9000000000 | -632.1224191760 | 82.39365367 |
| 1.0000000000 | -632.1460481500 | 20.35578243 |
| 1.1000000000 | -632.1535070430 | 0.772458857 |
| 1.2000000000 | -632.1538012570 | 0 |
| 1.3000000000 | -632.1515762760 | 5.841687616 |
| 1.4000000000 | -632.1490196910 | 12.55400153 |
| 1.5000000000 | -632.1470179040 | 17.8096933 |
| 1.6000000000 | -632.1457415820 | 21.16067671 |
| 1.7000000000 | -632.1447845590 | 23.6733406 |
| 1.8000000000 | -632.1429342760 | 28.53125862 |
| 1.9000000000 | -632.1375919840 | 42.55744626 |
| 2.0000000000 | -632.1238545320 | 78.62512649 |
| 2.1000000000 | -632.0932221670 | 159.0504008 |
HMim-CL PES Plot for Me3NH-Cl
Plot of Total relative energy (kJ/mol) vs Scan coordinate (Â)
This graph shows that when the N-Cl distance is set at 3.2 Â a broad minima occurs, the most stable state is an ion-pair Me3NH+ --- Cl- and as the proton is pushed to the Cl forming a neutral pair Me3N --- HCl the energy goes up, and no stable minima forms, rather there is a "shelf" in the PES. The ion-pair forms a doubly ionic H-bond between the Me3NH+ and Cl-, the neutral-pair forms a normal H-bond between the Me3N and HCl.
1-methyl-imidazolium chloride A
Calculation data
| name of submitted log file | IR_IMIDA_A_OPT.LOG |
| molecule | 1-methyl-imidazolium chloride A |
| method | RB3LYP |
| basis set | 3-21G(d,p) |
| final energy | -722.687898 |
| RMS gradient | 1.96e-07 |
| point group | C1 |
Item Table
Item Value Threshold Converged? Maximum Force 0.000000 0.000015 YES RMS Force 0.000000 0.000010 YES Maximum Displacement 0.000004 0.000060 YES RMS Displacement 0.000001 0.000040 YES
Low frequencies data
| Low frequencies | -5.1397 | -2.7750 | 0.0021 | 0.0028 | 0.0030 | 2.7889 |
| Low frequencies | 36.1426 | 64.4146 | 80.7977 |
Optimised molecule image
logfile: Media:IR IMIDA A OPT.LOG
Important Geometric parameters
Optimised bond lengths
r(H-N)(3-5)= 1.1776 Â
r(H-Cl)(3-14)= 1.7193 Â
r(N-Cl)(5-14)= 2.8914 Â
r(C-H)(1-2)= 1.07478 Â
r(C-H)(12-13)= 1.07388 Â
r(N-C)(5-12)= 1.3895 Â
r(C-N)(5-1)= 1.33207 Â
Scan for 1-methyl-imidazolium chloride A
Raw data
Data Table
| Scan Coordinate  | Total Energy (Hartrees) | Relative Total Energy kJ mol -1 |
| 0.8000000000 | -722.5958117430 | 219.3908443 |
| 0.9000000000 | -722.6503978370 | 76.07505448 |
| 1.0000000000 | -722.6727650690 | 17.34988686 |
| 1.1000000000 | -722.6793732910 | 0 |
| 1.2000000000 | -722.6792906320 | 0.217021205 |
| 1.3000000000 | -722.6772128850 | 5.672145953 |
| 1.4000000000 | -722.6753243630 | 10.63046046 |
| 1.5000000000 | -722.6744432310 | 12.94387253 |
| 1.6000000000 | -722.6746286640 | 12.45701819 |
| 1.7000000000 | -722.6753546960 | 10.55082117 |
| 1.8000000000 | -722.6753493340 | 10.5648991 |
| 1.9000000000 | -722.6721372800 | 18.99814688 |
| 2.0000000000 | -722.6613011090 | 47.44851384 |
| 2.1000000000 | -722.6354465340 | 115.3297005 |
Formal Scan Graph for 1-methyl-imidazolium chloride A
1-methyl-imidazolium chloride B
Calculation data
| name of submitted log file | IR_IMIDA_B_OPT.LOG |
| molecule | 1-methyl-imidazolium chloride B |
| method | RB3LYP |
| basis set | 3-21G(d,p) |
| final energy | -722.6662 |
| RMS gradient | 3.291e-06 |
| point group | C1 |
Item Table
Item Value Threshold Converged? Maximum Force 0.000006 0.000450 YES RMS Force 0.000002 0.000300 YES Maximum Displacement 0.000443 0.001800 YES RMS Displacement 0.000078 0.001200 YES
Low frequencies data
| Low frequencies | -5.0731 | -2.6203 | -0.0031 | -0.0021 | 0.0024 | 1.7049 |
| Low frequencies | 45.4247 | 162.0729 | 198.7836 |
Optimised molecule image and Jmol rotateable molecule
logfile: Media:IR IMIDA B OPT.LOG
Important Geometric parameters
Optimised bond lengths
r(H-N)(7-3)= 1.10445 Â
r(H-Cl)(7-14)= 2.13423 Â
r(H-Cl)(13-14)= 2.27699 Â
r(N-Cl)(8-14)= 3.64968 Â
r(C-H)(3-7)= 1.10446 Â
r(C-H)(10-13)= 1.10623 Â
r(N-C)(8-10)= 1.496 Â
r(C-N)(3-8)= 1.40206 Â
1-methyl-imidazolium chloride C
Calculation data
| name of submitted log file | IR_IMIDA_C_OPT.LOG |
| molecule | 1-methyl-imidazolium chloride C |
| method | RB3LYP |
| basis set | 3-21G(d,p) |
| final energy | -761.77953 |
| RMS gradient | 2.192e-06 |
| point group | C1 |
Item Table
Item Value Threshold Converged? Maximum Force 0.000006 0.000450 YES RMS Force 0.000002 0.000300 YES Maximum Displacement 0.000110 0.001800 YES RMS Displacement 0.000036 0.001200 YES
Low frequencies data
| Low frequencies | --3.6719 | -3.0923 | -0.0036 | -0.0031 | -0.0022 | 2.2057 |
| Low frequencies | 52.1748 | 102.5801 | 107.1376 |
Optimised molecule image
logfile: Media:IR IMIDA C OPT.LOG
Important Geometric parameters
Optimised bond lengths
r(H-C)(4-1)= 1.11785 Â
r(H-Cl)(4-17)= 2.03013 Â
r(H-Cl)(11-17)= 2.41692 Â
r(N-Cl)(7-17)= 3.66575 Â
r(C-H)(9-11)= 1.10140 Â
r(N-C)(7-9)= 1.49021 Â
r(C-N)(1-7)= 1.34794 Â
Ionic liquid ion pair (1-methyl-imidazolium chloride (HMim-Cl) A, B, and C)
Optimised H--Cl bond lengths
| Molecule Name | HMim-Cl A | HMim-Cl B | HMim-Cl C | Me3NH-Cl |
| Bond distance (Â) | 1.7193 Â | 2.13423 Â | 2.03013 Â | 1.73817 |
| Additional H--Cl bond | N/A | 2.27699 Â | N/A | N/A |
how do the H-bonds of Me3NH and HMim compare?
how do the H-bonds of the N-H and C-H H-bonds compare?
are these distances representative of a H-bond? Hint compare your distances to the sum of the van der waals radii of H and Cl.
will the ionic nature of the ions effect a distance based assessment of H-bonding?
Associated energies from 1-methyl-imidazolium chloride (HMim-Cl) A, B, and C
Optimised Ion and rotatable molecule for 1-methyl-imidazolium chloride (HMim-Cl) A & B
 |
===Jmol rotateable molecule===
logfile: Media:IROBERTSON_NH3_OPTF_POP.LOG
|
logfile: Media:IR_IMIDA_A&B_OPT_ION.LOG
Optimised Ion for 1-methyl-imidazolium chloride (HMim-Cl) C
logfile: Media:IR_IMIDA_C_OPT_ION.LOG
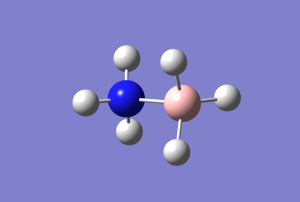
NH3 Molecule
Calculation data
| name of submitted log file | IRobertson_nh3_optf_pop.log |
| molecule | NH3 |
| method | RB3LYP |
| basis set | 6-31G(d,p) |
| final energy | -56.557769 |
| RMS gradient | 1.53e-07 |
| point group | C3v |
Item Table
Item Value Threshold Converged?
Maximum Force 0.000000 0.000015 YES
RMS Force 0.000000 0.000010 YES
Maximum Displacement 0.000003 0.000060 YES
RMS Displacement 0.000001 0.000040 YES
Low frequencies data
| Low frequencies | -5.6864 | -3.6131 | -3.6124 | 0.0017 | 0.0048 | 0.0162 |
| Low frequencies | 1089.3674 | 1693.9284 | 1693.9284 |
Optimised molecule image
Jmol rotateable molecule
logfile: Media:IROBERTSON_NH3_OPTF_POP.LOG
optimised NH molecule |
Important geometric parameters
Optimised bond distance and angle for NH3
r(N-H)=1.02Â
θ(H-N-H)=106°
Vibrational data
| Mode | 1 | 2 | 3 | 4 | 5 | 6 |
| Wavenumber (cm-1) | 1089 | 1694 | 1694 | 3461 | 3590 | 3590 |
| Symmetry | A1 | E | E | A1 | E | E |
| Intensity (arbitrary) | 145 | 14 | 14 | 1 | 0 | 0 |
Optimised molecule image with charges
| Atoms | N | H |
| Charge | -1.13 | 0.38 |
N2F2 Molecule
Calculation data
| name of submitted log file | IJR_N2F2_C2V_OPTF.LOG |
| molecule | N2F2 |
| method | RB3LYP |
| basis set | 6-31G(d,p) |
| final energy | -309.01241 |
| RMS gradient | 3.17e-07 |
| point group | C2V |
Item Table
Item Value Threshold Converged? Maximum Force 0.000000 0.000015 YES RMS Force 0.000000 0.000010 YES Maximum Displacement 0.000001 0.000060 YES RMS Displacement 0.000000 0.000040 YES
Optimised N2F2 molecule
Optimised N2F2 molecule image
Jmol rotatable molecule
logfile: Media:IJR_N2F2_C2V_OPTF.LOG
optimised NF molecule |
Important geometric parameters
Optimised bond distance and angle for N2F2
r(N-F)=1.39Â
r(N-N)=1.22Â
θ(F-N-N)=114°
θ(F-N-N-F)=0°
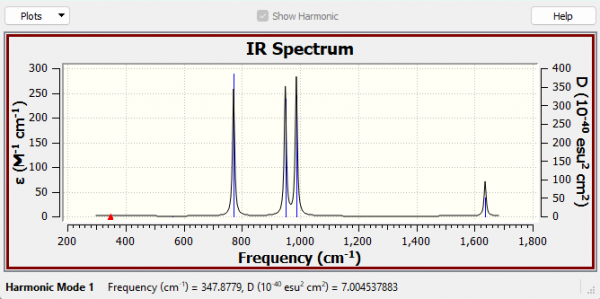
Vibrational Data and analysis
| Mode | 1 | 2 | 3 | 4 | 5 | 6 |
| Wavenumber (cm-1) | 348 | 561 | 772 | 949 | 987 | 1637 |
| Symmetry | A1 | A2 | B2 | A1 | B2 | A1 |
| Intensity (arbitrary) | 1 | 0 | 75 | 75 | 81 | 21 |
Which vibration is the asymmetric N-F stretch?
Vibration 3 is asymmetric.
What is the nature of the highest energy vibration?
It is an N-N stretching vibration
N2F2 Spectrum
IR Analysis
How many vibrations are expected from the 3N-6 rule?
We would expect there to be 6 vibrations (3(4) - 6 = 6)
Why are there only 4 peaks in the IR spectrum?
The A1 347.88 cm-1 and the A2 561.25 cm-1 modes have essentially no intensity, as both of these modes must have no dipole change. Therefore only the remaining 4 modes can be observed in the IR spectrum as peaks.
Low frequencies data
| Low frequencies | -0.0024 | -0.0016 | -0.0014 | 3.3364 | 4.3775 | 5.1348 |
| Low frequencies | 347.8779 | 561.2478 | 771.6088 |
Optimised molecule image with charges
| Atoms | N | F |
| Charge | 0.26 | -0.26 |
Molecular Orbital analysis
N2F2 MO 9
Which MOs are core orbital MOs?
The core MOs are the first four MOs 1-4 with low lying energy.